Poor UTI guidelines are driving AMR in women





The partnership between Forte Medical and Owen Mumford USA was launched today at the OCTANe Medical Innovation Technology Forum (MTIF) at Newport Beach, CA 28-29 October… Read More

A rise in persistent… Read More

New evidence showing a 70% reduction in false positive dipped specimens Extended uptake by the NHS Investment including a JV to create a Smart Peezy… Read More
Today, Forte Medical’s Chief Executive Giovanna Forte attended the Life Sciences Industrial Strategy reception at No 10 Downing Street – creating the foundation for brilliant… Read More

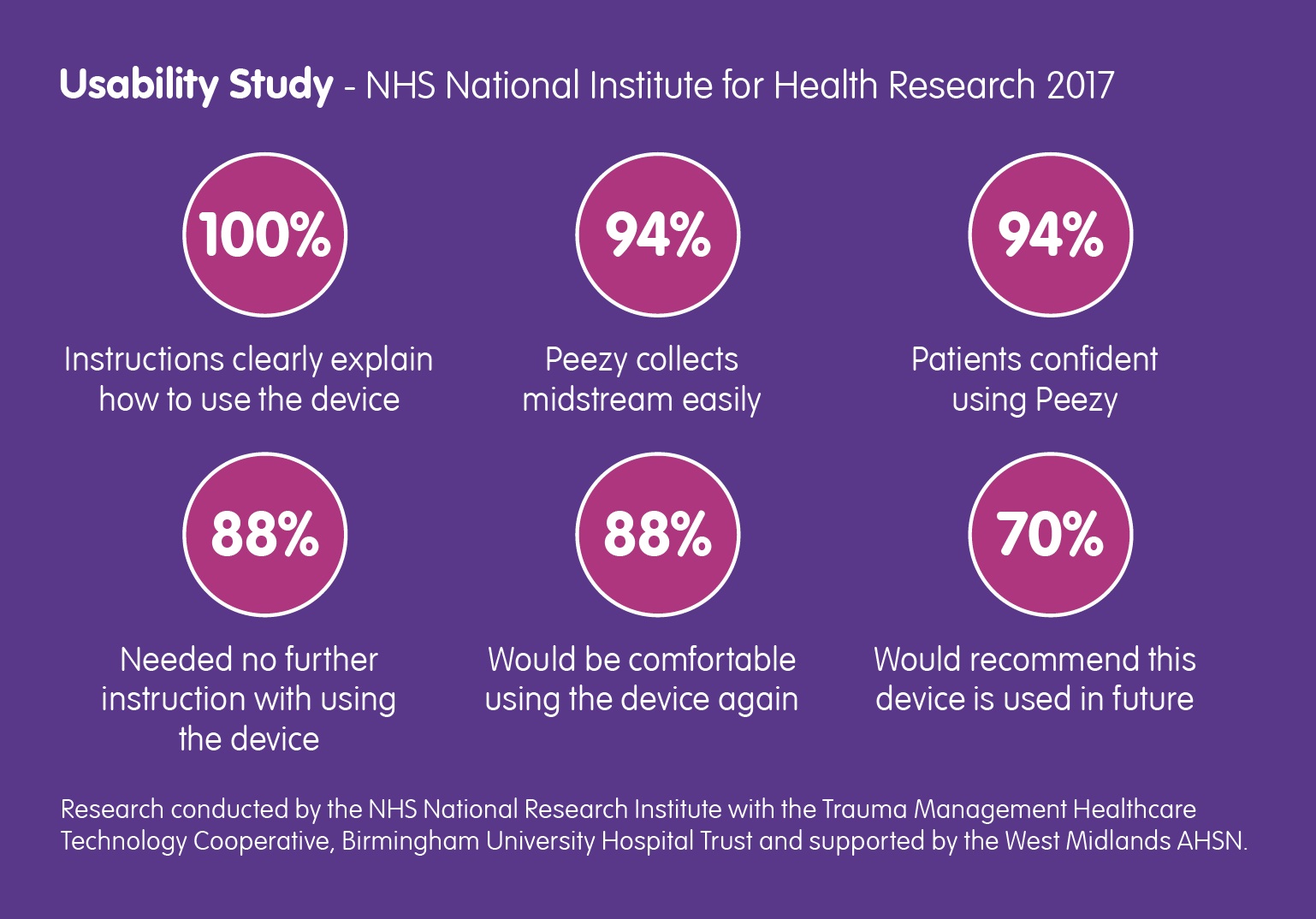
Back in March of this year, the NHS National Institute for Health Research contacted Forte Medical, asking for a case study… Read More
Interesting research being undertaken by Barbara Guinn, Head of Biomedical Sciences, University of Hull in The Conversation.com. “A urine test for ovarian cancer could… Read More
